A Guide to Medication Management Under the NDIS
Published: 30 August 2021

Published: 30 August 2021

Medications are substances that are given in order to prevent, diagnose, cure, control or alleviate disease, or improve a person’s physical or mental wellbeing. Medications can be prescription and non-prescription and include complementary healthcare products (APAC 2006).
Medications are crucial in maintaining health, preventing illness and treating health conditions - but when used inappropriately or incorrectly, they pose a serious risk of harm (TAS DoHHS 2016).
For this reason, workers employed by National Disability Insurance Scheme (NDIS) providers must be able to competently manage medications for participants who require them.
Medication management takes place at both an individual level and a system level. It may include:
(TAS DoHHS 2016)

Medications come in a variety of forms. Workers may need to administer:
(NDS 2016)
Management of medication is a requirement of the NDIS Practice Standards, under Core Module 4: Provision of Supports Environment.
This Practice Standard aims to ensure that NDIS participants feel confident that their provider administers, stores and monitors the effects of the medications being administered, and that providers work to prevent errors and incidents (NDIS 2020).
NDIS providers must meet the following quality indicators:
(NDIS 2020)
Medication Administration Records (MAR) are used to monitor, review and reconcile a person’s medication information and administration (TAS DoHHS 2016).
MARs are essential in reducing medication errors and incidents. They also:
(TAS DoHHS 2016)
A participant’s MAR should be a current, accurate and reliable record of all their medications. This should include all prescription, non-prescription, complementary and alternative medications being used by the participant (TAS DoHHS 2016).
NDIS Providers must ensure that MARs clearly state each participant’s required medicines and dosages. Information that is required to correctly identify participants and safely administer medicines must also be included (NDIS 2020).
In order to safely administer medicines, all workers must adhere to the following checking process known as the 13 Rights of Medication Administration:
(Ausmed 2019)
All medication administration should be documented (TAS DoHHS 2016).
Before administering medication, a worker should, as far as possible, understand:
(TAS DoHHS 2016)
This information can be provided by the participant’s pharmacist directly or via written information, for example, a consumer medicine information (CMI) leaflet.
CMI leaflets contain information on the safe and effective use of prescription and pharmacist-only medications. They provide information aimed at bringing about better health outcomes.
For more information on medications, search the NPS MedicineWise website.
A medication incident is ‘any event where the expected course of events in the administration of medications is not followed’ (TAS DoHHS 2016).
Medication incidents include:
(TAS DoHHS 2016)
Note: The following is intended as a guide only. Always refer first to your organisation’s policies and procedures for the most appropriate course of action.
(TAS DoHHS 2016)
All medications must be safely and securely stored in a way that maintains the quality of the medication and protects those who live in, work in or visit the service environment (TAS DoHHS 2016).
Wherever possible, dispensed medications should be kept in their original packaging (TAS DoHHS 2016).
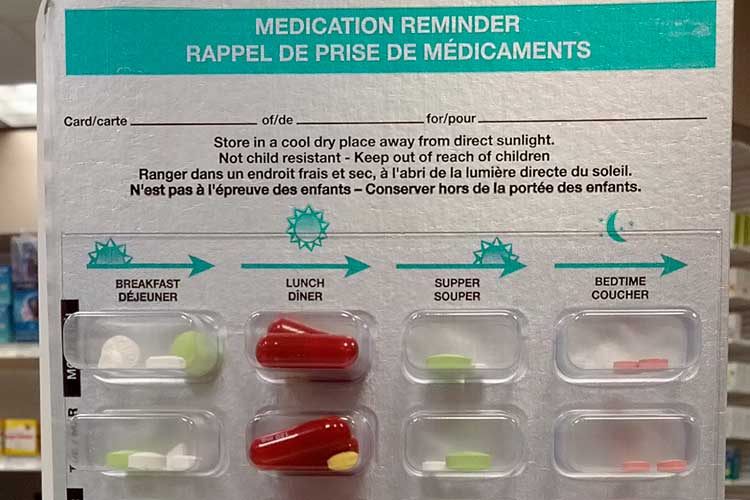
In some cases, a secure dosage administration aid (SDAA) might be deemed more appropriate for safely storing a medication (TAS DoHHS 2016).
An SDAA is a pharmacy-prepared aid that divides solid oral medications into sealed individual doses. The medications are arranged according to the dose schedule throughout the day (TAS DoHHS 2016).
Medications, including those in an SDAA, should be stored:
(TAS DoHHS 2016)
Keys to medication cabinets should be clearly labelled and kept by the personnel in charge of the premises (TAS DoHHS 2016).
In most cases, medications should be stored in a cool, dark, dry and secure place. However, certain medications may require special conditions, for example, refrigeration (TAS DoHHS 2016).
Medications should always be under the control of a specified worker. At the end of each shift, this worker should be responsible for checking the participant’s MAR to ensure that no medication administrations have been missed and that an accurate record has been kept (TAS DoHHS 2016).
Workers must always check the medication name on the packaging before beginning to prepare the medication for administration. The medication name on the packaging must be exactly the same as the medication name on the participant’s MAR and should be easily identified and differentiated by the active ingredient and medication strength.
Note that medications can have up to three different names:
| E.g. | Aspirin |
| Chemical name: | Acetylsalicylic acid |
| Generic name: | Aspirin |
| Trade name: | Aspro, Disprin, Astrix, etc. |
For this reason, it is advised to use generic names rather than brand names.
Medications might come in a box, bottle or another kind of packaging. They could also be packed in a pharmacy-issued SDAA, i.e. a blister pack or sachet. Medications that are not in either their original packaging, suitable packaging as supplied by a pharmacist or in a pharmacy-prepared SDAA should not be administered as they cannot be easily identified or differentiated. This means the potential for error is high and it is not possible to comply with the 13 rights of medication administration.

Disability service providers should ensure the workers they employ have a level of competency to provide appropriate and safe medication management support to people living with disabilities. Workers should not administer medications until training has been completed and they are deemed to be competent by an appropriate training organisation (TAS DoHHS 2016).
Question 1 of 3
The following names all refer to the same medication. Which of the following names should be used?