Pulmonary Embolism: DVT to PE
Published: 12 July 2021
Published: 12 July 2021

Throughout history, pulmonary embolism (PE) has almost always been diagnosed on the autopsy table. In other words, it was considered a ‘terminal event’ (Doherty 2017).
Today, PE is seen as a modern-day medical dichotomy: we understand its minutia and yet, somehow, still fail to diagnose or tackle it appropriately.
Appropriately treated, pulmonary embolism need not live up to its historical reputation as a life-ender.
Australia experiences approximately 17 000 new cases of venous thromboembolism annually (Tran et al. 2019), with pulmonary embolism accounting for 4 out of 10 of these cases (Skinner 2013).
Although PE is a significant factor in mortality and morbidity, the latest Australian and UK statistics on deaths due to pulmonary embolism are much lower than in the United States.
According to the most recent available statistics, PE accounts for only 0.2% and 0.4% of all annual deaths in Australia and the UK respectively (Doherty 2017). These are far smaller mortality rates than seen in the US, where it’s estimated that up to 100 000 people die from DVT/PE every year (CDC 2020a).
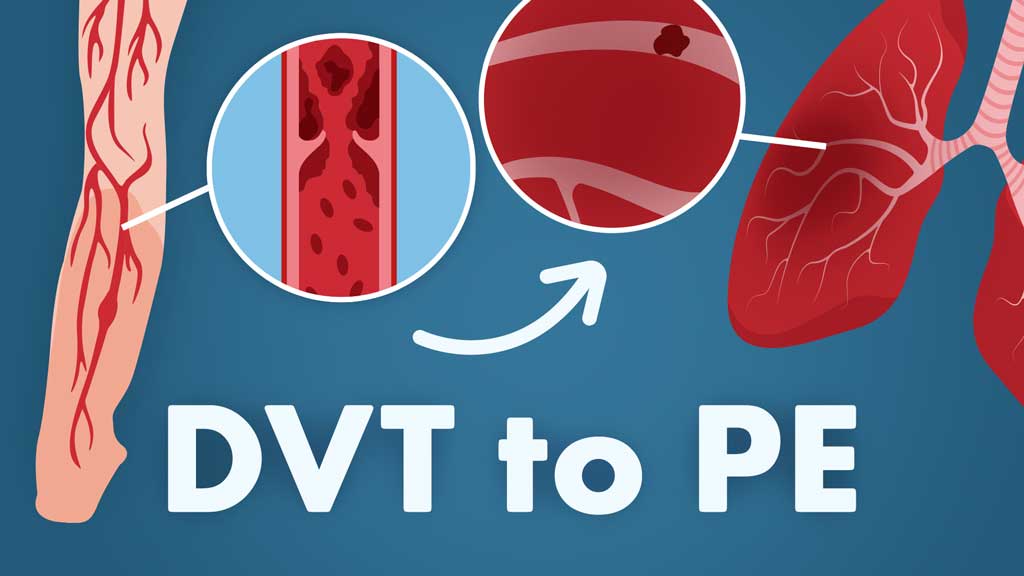
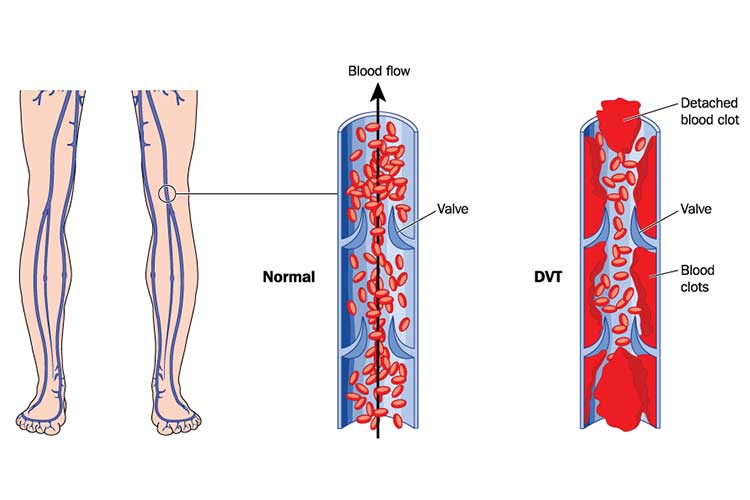
Pulmonary embolism (PE) is a natural consequence of a cascade of unfortunate events that begin with a thrombus formation, typically in a deep vein of the legs, thighs or pelvis (Fig. 1).
With venous thromboembolism events, where there is smoke, there is usually fire. Three risk factors, collectively described as the Virchow triad, are the ‘smoke’ that occurs prior to a PE diagnostic fire.
Virchow's triad consists of:
(Garmo, Bajwa & Burns 2020; Kushner, West & Pillarisetty 2020; Ausmed 2021)
The presence of these risk factors places patients at a significantly elevated risk of the venous event turning into a much bigger pulmonary problem.
In fact, over 50% of patients with a DVT will eventually end up with a diagnosis of PE (Merli et al. 2017).
It is not enough for clinicians to eyeball a patient’s clinical presentation to determine the need for medical imaging to rule out PE.
Instead, clinicians should use several valid and reliable checklists comprised of clinician characteristics in order to rule out the likelihood of PE. An example is this checklist by Kline et al. (2008):
A patient who presents with NONE of the following characteristics has a pretest probability of having a PE of less than 1%:
If the patient has at least one of the above characteristics, then PE cannot be ruled unlikely without further assessment.
Deep vein thromboses (DVT) can be caused by anything that prevents normal circulation in the legs, including bed rest or limited movement, injury, bracing, surgery, or even certain types of medicines (see risk factors for more info).
DVTs are largely preventable, which means that PEs are also largely preventable.
Patients who maintain a healthy weight, exercise (or at least do not maintain a sedentary lifestyle) and follow directions related to post-surgery care or bedrest are far less likely to develop a DVT.
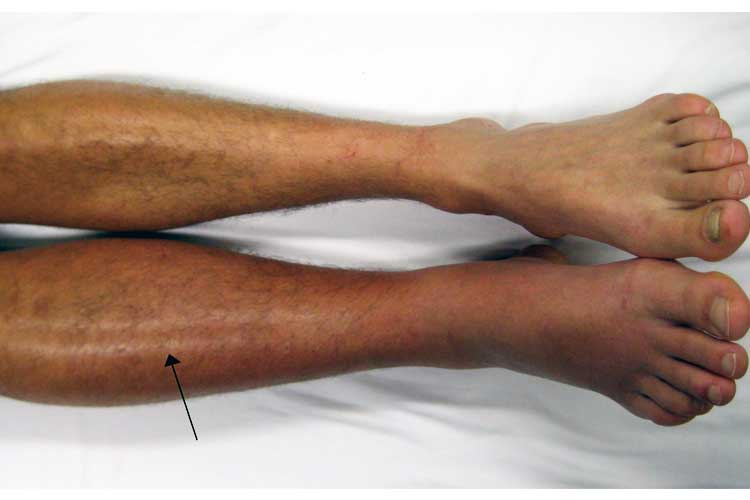
The most prominent signs of a DVT are:
A Homans’ sign is no longer considered a sensitive or specific test for DVT (Ambesh, Obiagwu & Shetty 2017).
View Full Lecture! →
Pulmonary embolism typically begins with the migration of a thrombus formation (Fig. 3), usually from the deep veins of the leg to the small blood vessels of the lungs. The clot lodges in these smaller pathways and blocks blood flow, potentially leading to tissue death.
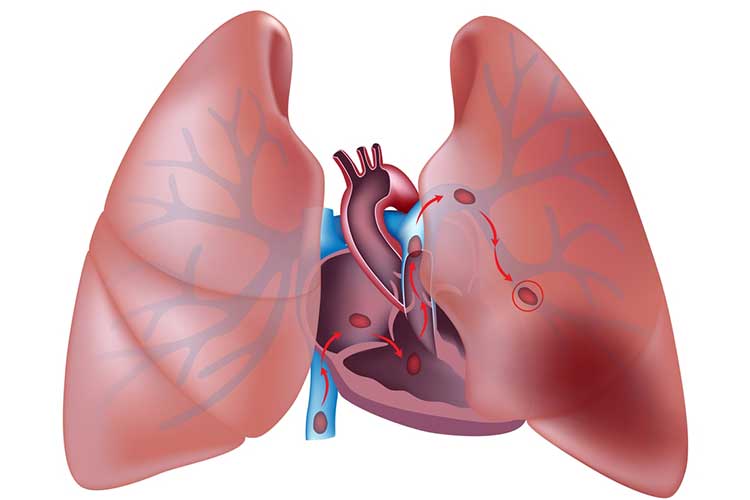
It is important to know that patients can suffer a PE without any prior symptoms of a DVT.
Signs and symptoms of PE can include:
(CDC 2020b)
Risk factors for DVT (and thus for PE) include many events that tend to lead to injuries to the venous structures of the leg, thigh, hip or pelvis.
Fractures, surgery and significant muscle tears are all conditions that create the potential for DVT formation.
Individuals with slow blood flow have an elevated risk; this flow problem can be due to prolonged sitting from long car rides or aeroplane flights, bed rest, or paralysis.
Pregnancy, hormone replacement therapy and contraceptive medicines are also major culprits due to a common theme: they cause an increase in oestrogen to be delivered to the body.
Central venous catheters must be closely monitored.
Even certain medical conditions such as cardiopulmonary disease , inflammatory bowel conditions and obesity can increase the risk.
Certainly, family history of DVT or PE or any type of genetic clotting disorder will also elevate the odds.
These odds increase further in older adults.
Over time, clinicians have developed working models to determine the probability of a PE diagnosis. There are many available, but the Wells model is easy to use and well established.
View the entire lecture, Recognising a Clot, and more from this conference:
| Active cancer (patient receiving treatment for cancer within 6 months or currently receiving palliative treatment) | 1 |
| Hemoptysis | 1 |
| History of deep venous thrombosis or pulmonary embolism | 1.5 |
| Surgery or bedridden for 3 days or more during the past 4 weeks | 1.5 |
| Heart rate > 100 beats/min | 1.5 |
| Pulmonary embolism judged to be the most likely diagnosis | 3 |
| Clinical signs and symptoms compatible with deep venous thrombosis | 3 |
(Streiff et al. 2016)
Individuals who score a high probability should skip the D-dimer assay and go immediately to imaging (CT angiography), which will either confirm or refute the presence of a PE.
Individuals with a moderate score should be tested with a high sensitivity D-dimer assay. If a high sensitivity D-dimer assay is not available, the patient should be imaged immediately with a CT angiography.
A computed tomographic pulmonary angiography (CTPA) is the standard diagnostic imaging tool used to determine the presence of a PE (Fig. 4). This test allows clinicians to visualise the small vessels of the lungs through the use of a contrast dye injected into a vein.
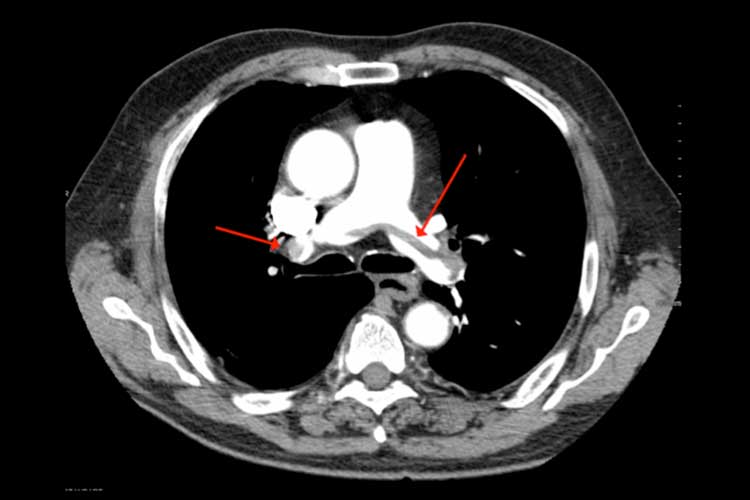
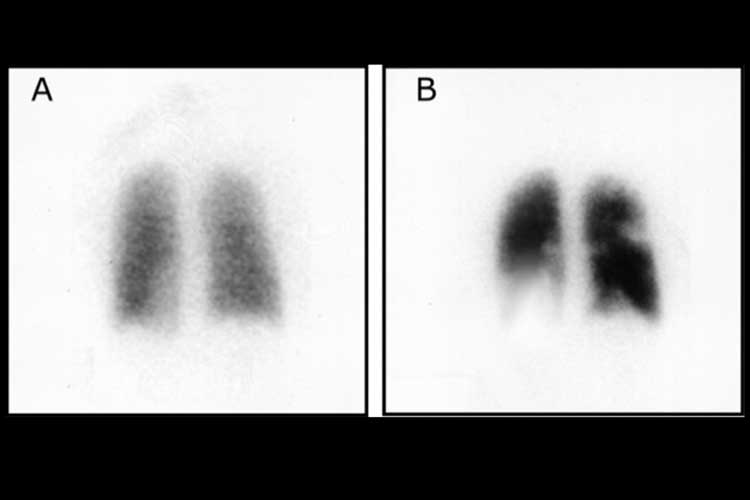
Other tests, such as ventilation-perfusion scans (Fig. 5) and pulmonary angiography, can also be used to diagnose PE.
Magnetic resonance imaging (MRI) can also be useful, but it is typically reserved for special populations where the use of radio waves is preferable to the use of X-rays, or where contrast dyes are not tolerated.
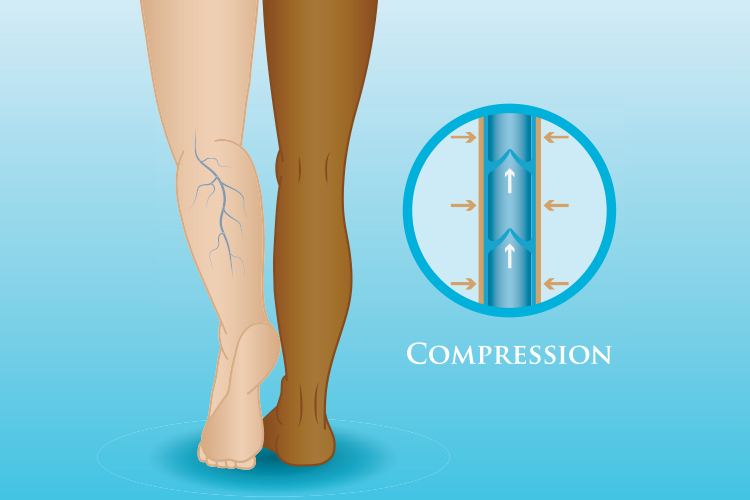
Compression stockings and vertical immersion in water (a swimming pool) are both great strategies designed to prevent DVTs and PEs (Fig. 6).
Medicine may be used for both the prevention and treatment of DVTs and PEs.
If properly prescribed, medicines such as anticoagulants and thrombolytics can nip the problem in the bud.
Anticoagulants are often referred to as ‘blood thinners’, but this is not actually what they do. Instead, they reduce the ability of blood to clot, allowing the body time to reduce the size of the clot by reabsorption. They also make it less likely that the blood will clot again in the near future.
All anticoagulants increase the likelihood of bruising and bleeding, however, so this means patients are required to remain under monitoring to determine that they have not over (or under) medicated.
Anticoagulants can be injected (e.g. heparin or fondaparinux) or swallowed (warfarin, dabigatran, apixaban, and others).
Thrombolytics work to dissolve clots. They are more aggressive than anticoagulants, with a greater likelihood of uncontrolled bleeding, so they are reserved for more severe situations (Streiff et al. 2016).
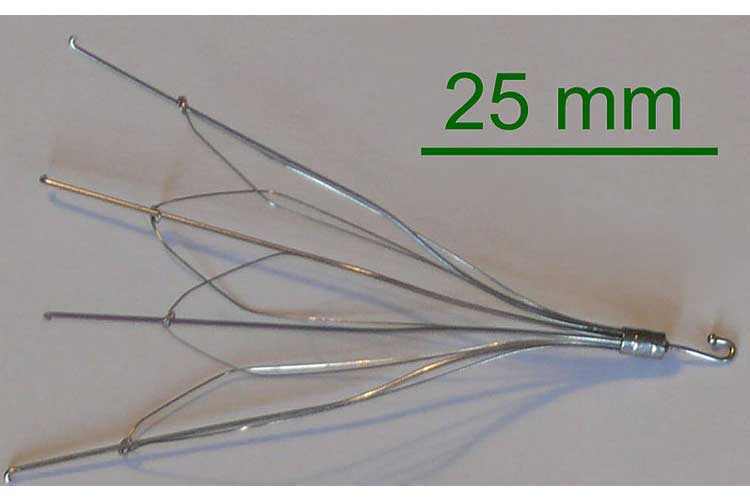
Two other management strategies are available if medicines do not do the job. A filter can be placed in the inferior vena cava in order to prevent any clots from migrating from the legs into the lungs (Fig. 7).
This is a prevention strategy and will do nothing after the fact. In contrast, thrombectomy and embolectomy are rare surgical procedures that are used to remove a clot in the legs or lungs, respectively (Streiff et al. 2016).
The first step to prevent PE is becoming aware of the risk. If you are at an increased risk, a doctor can recommend medicines or graduated compression stockings to help prevent a DVT from occurring.
As the risk of PE also increases when sitting for extended periods of time, this can be mitigated by getting up to walk, wearing loose-fitting clothes while travelling, and exercising your legs while seated.
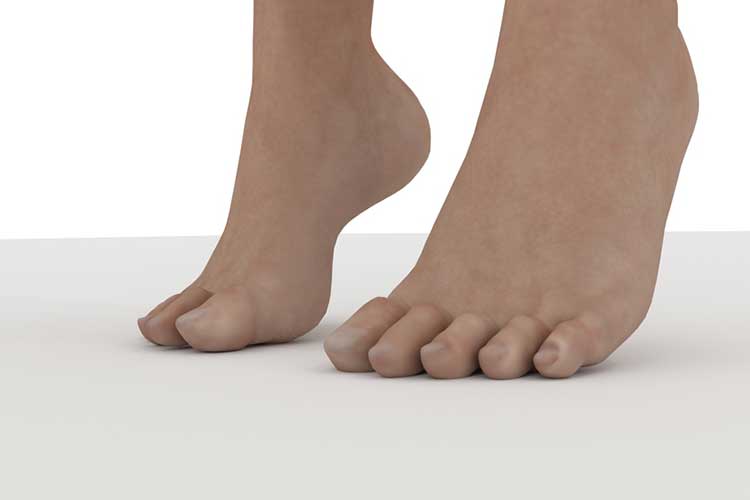
Stretching your leg muscles by lifting your heels off the ground and setting them down again (Fig. 8) is a good way to do this. Finally, maintaining a healthy weight and active lifestyle is key to reducing the risk of PE (Konstantinides et al. 2018).
Common complications include decreased quality of life, functional limitations, incomplete thrombus resolution, and abnormal pulmonary artery pressure and right ventricular function, among others.
OOne of the most severe complications, chronic thromboembolic pulmonary hypertension (CTEPH), is associated with a high risk of death. It occurs in just under 0.1% of post-PE patients but may be underdiagnosed due to presenting in a clinically similar way to PE (Guérin et al. 2014).
Identifying and correctly diagnosing CTEPH and other PE-related complications should be a primary concern of post-PE follow-up (D’Agostino et al. 2017).
As already discussed, the best clinical outcome is when the embolism never makes it to the lungs. And the best way to prevent the embolism from migrating to the lungs is to prevent it from ever forming in the lower extremities in the first place.
Clinician management of PE has changed over the years, with many excellent tools available to clinicians to allow them to bypass unnecessary tests and delay times.
With PE, time management is the most important treatment tool of them all.